Metabolic Flexibility: Why Humans Are Built for Both Plants and Meat
The body’s remarkable ability to shift between carbohydrate and fat-based metabolism as food environments change
Christopher Damman, Associate Professor of Gastroenterology, School of Medicine, University of Washington. Editor-in-Chief of Gut Bites MD.
Modern nutrition debates often frame diet as a choice between opposing camps: plant-based versus animal-based, carbs versus fat, glucose versus ketones. But evolution probably never forced humans to permanently choose one over the other. For most of human history, food availability shifted with the seasons. Wet seasons or summers often brought berries, tubers, honey, legumes, and fibrous plants, while dry seasons or winters likely pushed humans toward hunting, marrow, fat, and animal protein. Rather than existing in one stable nutritional state, humans probably moved back and forth between these environments repeatedly over thousands of generations.
This may explain why humans evolved such remarkable metabolic flexibility. Instead of specializing like strict carnivores or herbivores, we appear built to adapt. Our biology can shift fuel sources, digestive priorities, microbial activity, and even gene expression depending on what foods are available. In many ways, the defining feature of human metabolism may not be commitment to a single diet, but the ability to transition between them.
The Hadza Diet Offers a Glimpse Into the Past
One of the clearest modern examples comes from the diet patterns of the Hadza hunter-gatherers in Tanzania. Their food intake naturally changes throughout the year depending on rainfall, plant growth, and hunting success. During some seasons they consume large amounts of fibrous tubers, berries, baobab, and honey, while during other times meat intake rises substantially.
What makes this especially fascinating is that their microbiome changes alongside their diet. Certain microbial species expand during plant-rich seasons and then decline during more animal-based periods, only to return again when fibrous foods reappear. Their gut ecosystem itself appears metabolically flexible, adapting to changing food environments much like the human body does. This seasonal oscillation may resemble how humans ate for much of evolutionary history.
Our Cells Can Literally Run on Different Fuels
This flexibility is reflected deep within our biology. When carbohydrate intake is high, the body primarily relies on glucose for energy. But when carbohydrates become scarce and fat intake or fasting increases, the liver begins producing ketones like beta-hydroxybutyrate from fat. Remarkably, even the brain, one of the body’s most energy-demanding organs, can transition between these fuel systems.
That ability is extraordinary from an evolutionary perspective. Most tissues throughout the body, including muscle, liver, heart, and immune cells, can alter how they generate energy depending on what foods are available. Scientists refer to this as metabolic flexibility: the ability to efficiently switch between fuel sources rather than becoming locked into a single metabolic mode. This likely evolved because human food environments were rarely stable for long.
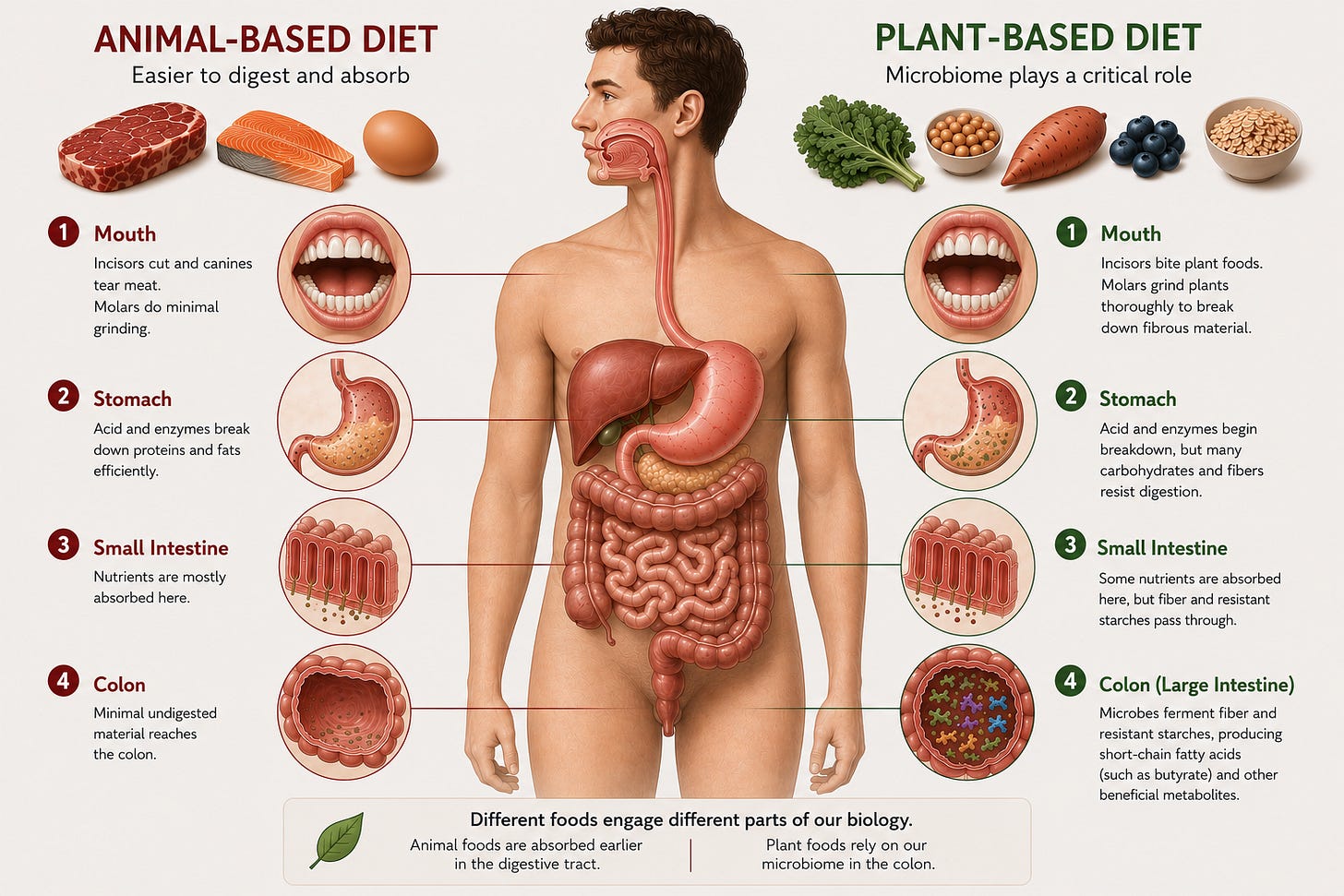
Plants and Animals Engage Different Parts of Our Biology
Plant-heavy and animal-heavy diets do not simply provide different calories. They activate different digestive and metabolic systems. Animal foods are relatively easy for the body to digest and absorb. Meat and fat are rapidly broken down by stomach acid and digestive enzymes, with much of their nutrients absorbed early in the small intestine.
Plants work differently. Plant cells are surrounded by fibrous cell walls that human digestive enzymes cannot fully break apart. As a result, substantial portions of plant material travel into the lower intestine, where gut microbes begin fermenting and transforming them. In many ways, a plant-rich diet recruits the microbiome as an additional metabolic organ.
These microbes help break down fiber and resistant starches into short-chain fatty acids like butyrate. They also transform polyphenols and other plant compounds into bioactive metabolites that humans may not efficiently produce on their own. While the microbiome likely plays a smaller role during heavily animal-based dietary periods, it becomes critically important during plant-rich periods where microbial fermentation helps unlock nutrients and signaling molecules from otherwise inaccessible plant material.
Carnivores, Herbivores, and Humans
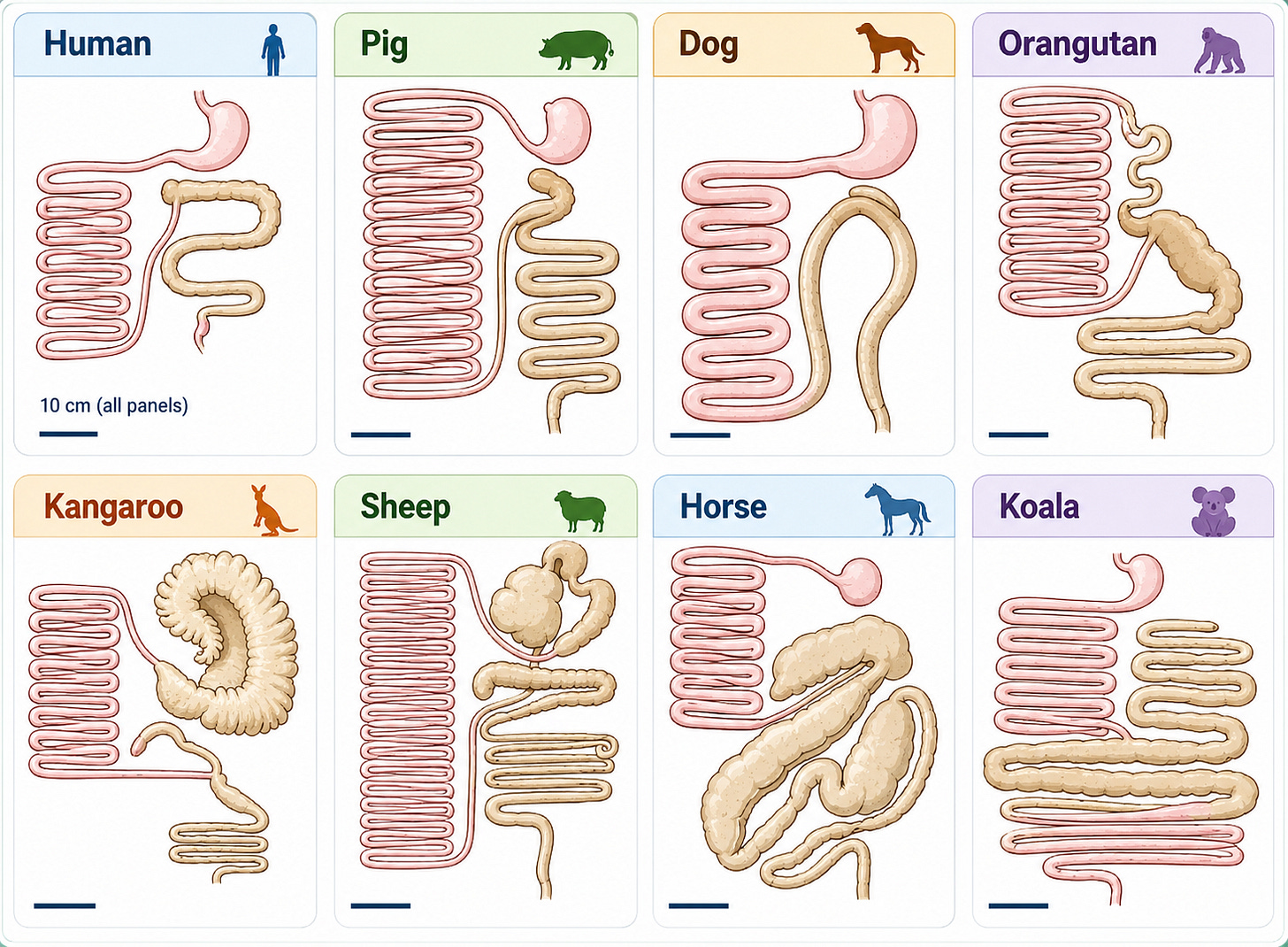
This difference is reflected across the animal kingdom. Carnivores generally have relatively short digestive tracts and small colons because meat is energy-dense and rapidly absorbed. Herbivores are the opposite. They often possess long intestines and large colons or foreguts, fermentation chambers designed to support extensive microbial breakdown of fibrous plants. Animals like cows rely heavily on microbial fermentation to extract energy from grasses humans could never digest on their own.
Humans appear built somewhere in the middle. Our digestive tract is longer than a carnivore’s but far shorter than a dedicated herbivore’s. Our teeth also reflect this mixed strategy. We have incisors and canines capable of tearing food, but also broad molars designed for grinding plants. As omnivores, we seem to possess elements of both systems, allowing us to shift between them depending on environmental conditions and food availability.
The Microbiome May Have Helped Bridge Nutritional Gaps
One fascinating possibility is that the microbiome helped humans obtain nutrients from plants that were otherwise more directly available in animal foods. Gut microbes can synthesize certain vitamins and unlock micronutrients trapped within plant structures. During seasons when meat was less available, microbial metabolism may have helped compensate for some of these nutritional differences.
But the microbiome may have done something even more important than nutrient extraction. It may have functioned as a signaling system that helped regulate metabolism itself. Many microbial metabolites do not simply provide calories. They act as molecular instructions that influence inflammation, mitochondrial activity, energy production, and gene expression.
One of the clearest examples is butyrate, a short-chain fatty acid produced when microbes ferment dietary fiber. Butyrate is not only a fuel source for colon cells. It also functions as an epigenetic signaling molecule that influences HDAC enzymes involved in metabolic regulation and mitochondrial function.
Butyrate and Beta-Hydroxybutyrate: Two Parallel Signals
What makes this especially interesting is that butyrate shares some functional similarities with beta-hydroxybutyrate, the ketone body produced during ketosis. At first glance they seem completely different. One emerges largely from plant fiber fermentation by microbes, while the other comes from fat metabolism during fasting or carbohydrate restriction.
Yet both molecules appear capable of influencing mitochondrial function, inflammation, oxidative stress, and epigenetic signaling pathways. In other words, both may help shift the body into a more stress-resistant and metabolically adaptive state. One pathway emerges from a fiber-rich plant environment, while the other emerges from fat metabolism and ketosis.
This kind of redundancy appears throughout biology. Evolution often preserves multiple ways to activate important survival pathways. Humans may have evolved overlapping systems that allowed periods of plant-rich microbial fermentation and periods of ketosis to both support metabolic resilience under different environmental conditions.
Why Ketogenic Diets May Sometimes Help
This broader evolutionary perspective may help explain why ketogenic diets can sometimes produce meaningful metabolic improvements, particularly in carefully supervised medical settings. Ketosis may temporarily activate ancient pathways associated with fasting, scarcity, cellular repair, and mitochondrial stress resistance. For some individuals, this metabolic reset may improve insulin sensitivity, appetite regulation, or neurological function.
But that does not necessarily mean humans evolved to remain in ketosis permanently. More likely, humans cycled between metabolic states depending on season, geography, migration, hunting success, illness, and food availability. Periods of lower carbohydrate intake may have alternated with periods rich in fibrous plants that nourished both humans and their microbiome.
That distinction matters. A metabolically restorative phase may not be the same thing as an ideal permanent state. Evolutionary health may have depended less on staying in one mode continuously and more on maintaining the flexibility to transition between them.
The Bigger Evolutionary Picture
Modern diets often remove the natural oscillations our biology may have evolved around. Ultra-processed foods provide continuous refined carbohydrates, concentrated fats, constant availability, and very little nourishment for the microbiome. Seasonal variation largely disappears, and the body may become metabolically locked into a narrow range of inputs.
But our biology still appears designed for variation. Variation in fuel sources. Variation in microbial activity. Variation in fasting and feeding. Variation in plant and animal intake. Rather than being optimized for one extreme dietary philosophy, humans may have evolved to move fluidly between nutritional environments depending on circumstance.
The goal may not be choosing one metabolic state forever. It may be preserving the flexibility to do both.
Christopher Damman, Associate Professor of Gastroenterology, School of Medicine, University of Washington. Editor-in-Chief of Gut Bites MD.